Bases des polymères
What is a „Polymer”?
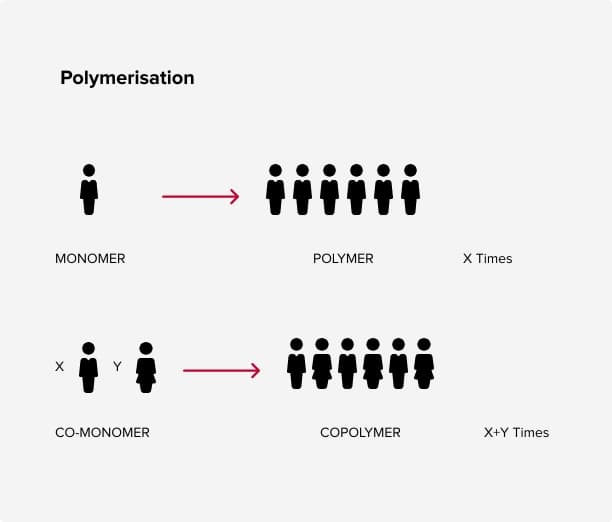
The word Polymer comes from the Greek ”poly” meaning many, and ”meros”, parts or units. A polymer is a group of many units. You combine many monomers (one unit) to create a polymer.
Polymer is often used as a synonym for ”plastic”, but many biological and inorganic molecules are also polymeric. All plastics are polymers, but not all polymers are plastics. Plastic actually refers to the way a material melts and flows.
Commercial polymers are formed through chemical reactions in large vessels under heat and pressure. Other ingredients are added to control how the polymer is formed and to produce the proper molecular length and desired properties. This chemical process is called ”polymerization”.
A homopolymer results from polymerizing only one kind of monomer. A copolymer results from using different monomers. Homopolymers have the same repeating unit while copolymers (which can be random, block or graft) can very have different numbers of repeating units. A terpolymer results from using three different monomers.
Materials for thermoforming
Thermoplastic materials can be repeatedly softened by elevated heating and hardened by cooling. They are either linear polymers or slightly branched. They consist of long molecules and each may have side chains or molecular groups not attached (not cross-linked). There are now newly developed thermoplastics that process as usual, but, at the end of the process are cross-linked with special techniques (e.g., nucleating agent, hot mold above forming temperature, etc.) Materials can also be across- linked by radiation. This cross-linking turns them into either an undeveloped or full thermoset and can greatly improve the physicals properties of the part.
Thermoplastics make up approximately 88-90% of all plastics processed today.
Any thermoplastic resin that can be extruded can be thermoformed. However, those with low hot strenght at forming temperature may be very difficult to form. Sheet and film is produced by extrusion, co extrusion, continuos casting, extrusion casting, calendring, compression molded, autoclave and press laminating, etc.
Basics
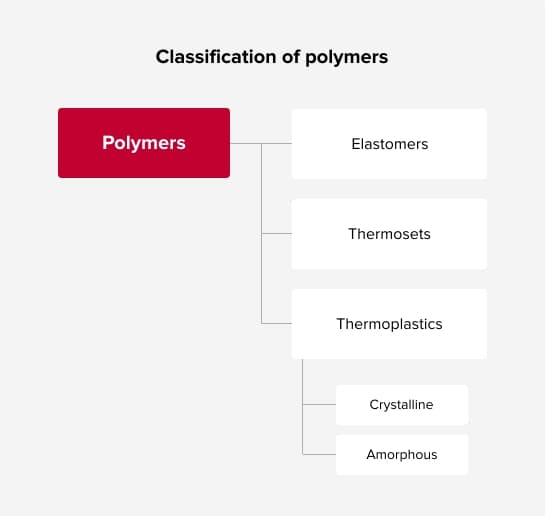
Elastomers are plastic material which can be expanded considerably by little force and which will return quickly to their original shape without force. There will be no plastic flow if temperature is increased as occurs with thermoplastics. Thermosettings plastics are hard, solid and remain in this state up to the decomposition temperature. Thermoplastics soften with increasing temperature after the softening point has been exceeded and harden again when cooled down. This procedure, which can be repeated as often as requested, is used for thermoforming of semi-finished products.
Thermosettings plastics are hard, solid and remain in this state up to the decomposition temperature.
Thermoplastics soften with increasing temperature after the softening point has been exceeded and harden again when cooled down. This procedure, which can be repeated as often as requested, is used for thermoforming of semi-finished products.
Representative skeletal structure of polymers
The structure of thermoplastics
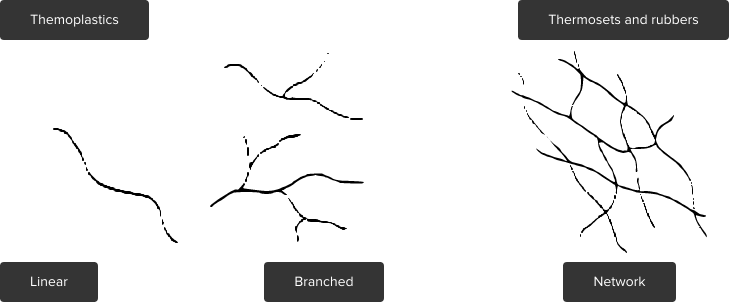
Thermoplastics are plastic materials which consist of linear or branched macromolecules (filamentary molecule).
With random convolution of the filamentary molecules one talks about amorpous thermoplastics.
If atomic groups of several linear molecule chains are arranged, they create a crystal lattice. So-called crystallites are formed and these integrate randomly into the amorphous substance.
Types of thermoplastics
There are two phases of Thermoplastics-amorphous and crystalline. Amorphous phase- Devoid of crystallinity; no definite order. Amorphous materials have a randomly ordered molecular structure. Its behavior is very similar to a very viscous, inelastic liquid. Upon heating, an amorphous sheet gradually softens and eventually acquires the characteristics of a liquid, but without a definite point of transition from solid to liquid state.
These resins usally require less energy to bring them to forming temperature and to cool they do not reach a totally "non-flowing" solid state. They do, therefore, have a tendancy toward "creep" or "movement" with age when a load is applied. Such plastics as the following are amorphous: ABS, HIPS, PVC, PMMA and PC are typical examples.
Structure of thermoplastics
Amorf

Crystalline

Crystalline Phase - A very good orderly group of molecules. Crystalline thermoplastics molecules have natural tendancy to line up in rigid, precise, highly-ordered structures but a part of the material will always be amorphous (e.g., polypropylene normally is about 65% crystalline and 35% amorphous). When a crystalline sheet is heated it remains stiff until it reaches the “transformation temperature”. For amorphous polymers this transformation temperature corresponds to Tg, but for crystalline polymers this temperature is slightly below the melt temperature.
This temperature is the minimum forming temperature and in case of HDPE it is 105degC. The ideal forming temperature is higher, but unfortunately, just some degrees below the melt temperature. This is why sheets from crystalline polymers are formed “cold”, which can result in internal stresses in the formed detail which in turn can lead to warpage. less impact strength, etc. This is why these materials are very difficult to thermoform. HDPE and PP (natural or filled) are the most usual types of crystalline sheet material.
Definitions
Organic Substance
Substance containing carbon
Polymeric Material
Material containing polymer as a main substance.Often containing different kinds of additives. Ex. Plastics, Rubber, Glue, Paint
Polymer
Poly = many, Mer = Part. Chemical, often organic,substance built up by chain shaped molecules.
Plastics Material
Polymeric materials which turns stiff after formingin a soft plastic state. Plastic = Polymer+Additives
Thermoplastic Material
Plastic Materials which can be melted and reshaped time after time
Elastic Material
Polymeric material that after fabrication in the plastic state turns soft with large elastic flexibility.
Thermosetting Plastic
Plastic Material which can not be reshaped.The material is decomposed during heating.